
You must think about the specific hazards and controls your business needs. the steps you need to take to manage the risksĭo not just copy an example and put your company name to it as that would not satisfy the law and would not protect your employees.You can use them as a guide to think about: These typical examples show how other businesses have managed risks. Risk assessment template (Open Document Format) (.odt).Risk assessment template (Word Document Format).what further action you need to take to control the risks We have been extensively discussed about the Failure Mode Effect Analysis as a tool to be used within the risk management process for medical device development.what you're already doing to control the risks.You can use a risk assessment template to help you keep a simple record of: Risk assessment template and examples Template The RMP template document also requires companies to elaborate on how they will be involved in on-going studies to minimize or prevent risks and enhance the. 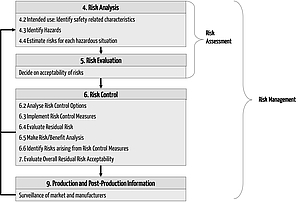
You can help us by completing a short survey. A Risk Management Plan or RMP is a document that details the medicinal product’s safety features, particularly the contingencies in place to reduce its risk severity amongst patients. You will learn how to answer difficult questions such as: Is my medical device safe. We want to find out more about who uses this part of our website and what they think of it. This book is about the management of safety risks for medical devices.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
